83% Sequential Net Revenue Growth to $54.2 Million
EDMONTON, Feb. 11, 2019 /PRNewswire/ – Aurora Cannabis Inc. (the “Company” or “Aurora“) (NYSE: ACB) (TSX: ACB) (Frankfurt: 21P; WKN: A1C4WM), announced today its financial and operational results for the second quarter ended December 31st, 2018.
Q2 2019 Financial and Operations Highlights
Q2 2019 and Subsequent Highlights
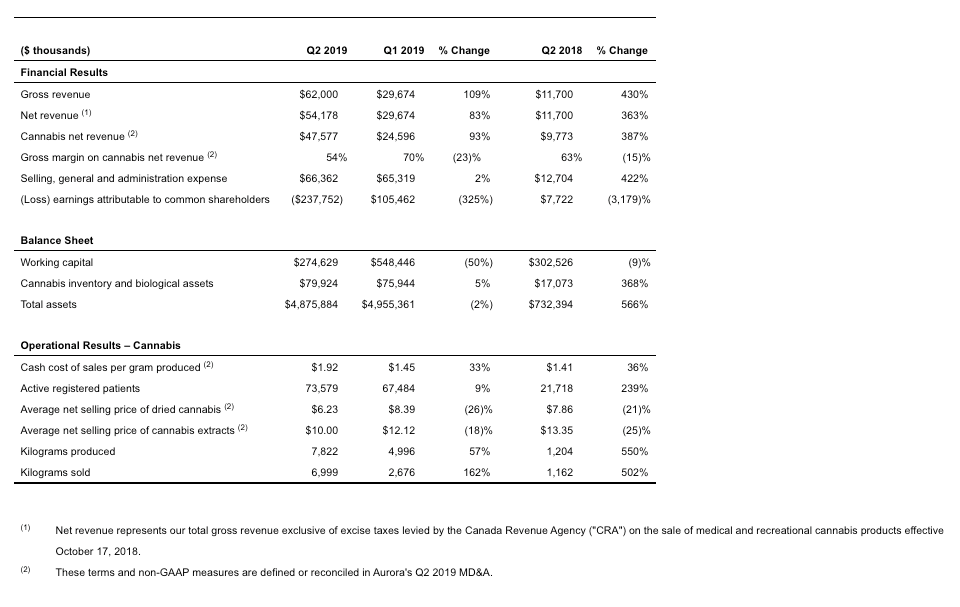
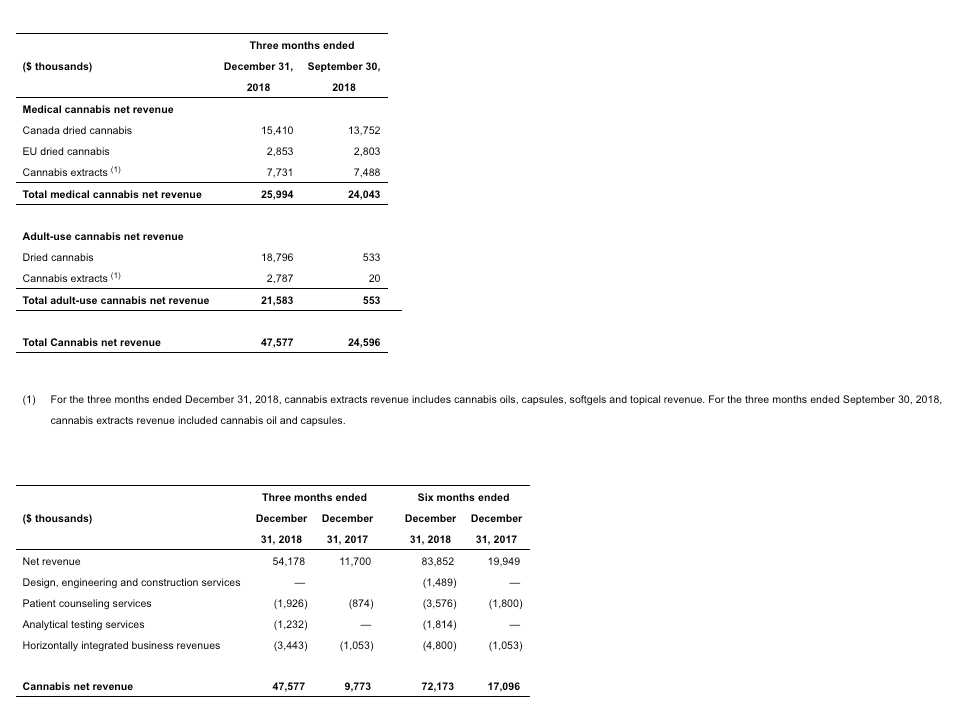
- Net revenue of $54.2 million, up 83% sequentially, and up 363% compared to the same period in 2018, driven by Aurora’s strong performance in the launch of the Canadian consumer market with sales of $21.6 million, and the Company’s continued strength in the Canadian and international medical markets with sales of $26.0 million, up 8% in revenue and 23% in volume sold.
- Average selling prices were impacted by the introduction of excise taxes across all Canadian sales channels on October 17, 2018, as well as lower wholesale pricing realized in the Canadian consumer market. Going forward, Aurora intends to continue prioritizing medical patients in Canada and globally where margins continue to exceed those achieved on the wholesale consumer market.
- Q2 2019 kilograms produced and kilograms sold of 7,822 and 6,999 were up 57% and 162%, respectively, driven by continued and significant scale-up of Aurora’s cultivation operations and strong demand across all the Company’s markets.
- Gross margin on cannabis sales of 54% was temporarily down from 70% in the prior quarter. The decrease was primarily due to a lower average selling price per gram of dried cannabis, the impact of excise taxes on medical cannabis net revenues, and a temporarily lower proportion of cannabis oil sales in the Company’s sales mix ratio. Also impacting gross margin were increased packaging requirements under the Cannabis Act and one-time ramp up and optimization costs as our Sky facility was brought up to full production. The Company anticipates that the launch of new derivative product lines, once allowed under Health Canada regulations, will contribute to improving margins.
- The company is performing well in the Canadian consumer market, recording $21.6 million of revenue in Q2 2019. Based on available data released by Health Canada for the Q2 2019 period, Aurora accounted for approximately 20% of all consumer sales across the country.
- Cash cost to produce per gram of dried cannabis sold temporarily increased from $1.45 in the previous quarter to $1.92 in Q2 2019. This change was primarily due to ramp-up and optimization costs as the Company scaled-up Aurora Sky to full production. One-time additional costs incurred related to the launch of the Canadian consumer market, as the Company waited for its Sky sales licence (received October 17, 2018), also contributed to the increase.
- Aurora Sky is now fully complete and commissioned, and is expected to reach its full production capacity, based on Health Canada approved planted rooms, shortly. Recent harvests completed to date at the facility have exceeded targeted yields, reflecting that the facility’s commissioning has been successful, all environmental and nutrition systems, and operating protocols are dialed in, and technology components are functioning well.
- Q2 2019 SG&A remained steady compared to the prior quarter as lower sales and marketing costs were offset by one-time public company and acquisition costs, as well as the absorption of a full quarter of SG&A costs from recently acquired companies, including MedReleaf.
- Non-cash expenses including the December 31, 2018 mark-to-market adjustments of approximately $190 millionprimarily on the Company’s derivative investments contributed significantly to a net loss of $240 million.
- In January 2019, Aurora completed a US$345 million convertible note offering, with the proceeds earmarked predominantly to drive the Company’s continued high pace of growth in Canada and internationally.
Outlook
The most significant driver of Aurora’s revenue growth over the next twelve to eighteen months is the Company’s scale-up of high-quality production available for sale to the Canadian consumer market and the Canadian and international medical markets. Aurora is now operating at an annualized production rate of approximately 120,000 kgs, based on Health Canada approved planted rooms, and expects to reach in excess of 150,000 kgs by March 31, 2019. Management reiterates previous guidance that based on the Company’s current confirmed production results, Aurora will have approximately 25,000 kgs available for sale in Q4 (April to June 2019).
The Company anticipates that with Aurora Sky operating at full capacity, as well as continued reduction in operating costs, the cash cost to produce per gram will trend significantly lower. Management reiterates its expectation that the sustainable long-term operating cost at its Sky Class facilities will be well below $1 per gram.
Ongoing disciplined cost management is expected to result in SG&A costs growing modestly as compared to revenue growth over the remainder of the fiscal year.
Consequently, and consistent with previous guidance, management believes that the combination of substantial revenue growth, low cost of production, and disciplined operating cost management will position Aurora to achieve sustained positive EBITDA beginning in fiscal Q4 2019 (calendar Q2 2019).
Longer term, the Company expects that the launch of new higher value-added derivative product lines in relation to anticipated changes in Health Canada regulations, as well as the introduction of derivative products to international markets, will contribute to further revenue growth and margin expansion.
Management Commentary
“Aurora continues to execute strongly across all of its market segments, as demonstrated by the 83% revenue growth over last quarter and the significant increase in confirmed production results,” said Terry Booth, CEO of Aurora. “Our brands continue to resonate extremely well in the consumer market, our patient numbers continue to increase steadily, and we have maintained our market leadership in Germany and other key international markets. We are experiencing exceptional demand for our Canadian medical and consumer products, as well as sustained strong demand internationally. With our Aurora Sky and MedReleaf Bradford facilities ramping up production as anticipated and our other licensed facilities operating at full capacity, we are reiterating our earlier guidance of achieving sustained EBITDA positive results from the second calendar quarter of this year (our fiscal Q4).”
Glen Ibbott, CFO of Aurora added, “We are also very pleased with our recent placement of US$345 million in convertible notes. These convertible notes were subscribed by high-quality US, Canadian, and international institutions and offer Aurora the flexibility and optionality to settle the entire principal amount of the notes in the future for cash, shares, or any combination thereof. This funding sufficiently supports the global opportunity for us to continue our commitment to growth in the legal, regulated medical and consumer cannabis systems across the globe. This is a unique time and position as we maintain a high cadence of increasing product supply and international market expansion.”
Mr. Booth concluded, “With our strong performance in the Canadian medical and consumer markets, our early mover advantage in a growing list of important international markets, together with our leadership in high-quality, CBD-rich hemp production, Aurora is strategically positioned across the entire cannabis industry value chain to further extend our rapid growth.”
Q2 2019 and Subsequent Operational Highlights
International Expansion
As the global cannabis market continues to mature, Aurora has secured a distinct first-mover advantage by attaining valuable import and export agreements with markets that have a high barrier to entry, reinforcing the Company’s commitment to serving the global cannabis industry. New supply agreements this quarter have expanded the Company’s sales and operations to 23 countries.
- Exports to Poland In October 2018, Aurora Deutschland GmbH shipped products to a pain treatment center and hospital in Warsaw, making Aurora’s high-quality medical products available for Polish patients. This shipment is believed to be the first time a non-government run business has been granted approval to supply medical cannabis products in the country.
- Exports to Czech RepublicDuring Q2, the Company secured export permits and completed its first shipment of medical cannabis to Czech Medical Herbs s.r.o. (“CMH”), a Czech pharmaceutical wholesaler.
- Export to Luxembourg In December 2018, the Company was selected by the Luxembourg Health Ministry for the supply of medical cannabis to the country and an initial purchase order for approximately 20 kilograms was received from Luxembourg officials. To date, Aurora is the only company supplying the Luxembourg medical cannabis system.
- Exports to MexicoOn December 7, 2018, prior to Aurora entering into a Letter of Intent to acquire Farmacias, the Company entered into discussions with the Mexican pharmaceutical manufacturer to import Aurora THC products for distribution to the Mexican medical cannabis market. The Company believes that Farmacias is the only company in Mexico with the required licenses to import and distribute products with a THC content greater than 1%.
- Exports to United KingdomOn February 11, 2019, the Company announced that it had completed its first commercial export of cannabis oil to the United Kingdom (the “UK”), which was subsequently dispensed from a pharmacy. UK authorities have recently granted Aurora approval for its first shipment under the new legal framework that came into effect November 1, 2018 and the Company is enhancing its UK operations to ensure patient access to a range of cannabis-based medicines.
Product Developments
During the quarter, the Company maintained its focus on product development and continued to execute on its strategy to generate a broad portfolio of high-margin, targeted medical and consumer products.
- Launch of Canada’s First Legal Vape-Ready CBD Oil Cartridges On October 16, 2018, Aurora received all necessary compliance verifications from Health Canada to commence sales of the first product from the Company’s innovative, high-potency, vape-ready CBD oil product line, Aurora Cloud. To date, Aurora Cloud products are the only vape-ready CBD products legally available in Canada. The initial product release is the first of a broader line of full-spectrum cannabinoid-based, vape ready cartridge products, which the Company intends to launch in the near future.
- Launch of Softgel CapsulesOn December 3, 2018, the Company commenced shipments of cannabis softgel capsules to the Canadian medical and consumer markets from its state-of-the-art Aurora Vie facility. As an alternative drug delivery method, Aurora intends to make this high-volume, smoke-free product available to key domestic and international markets where legally permissible.
- Ongoing Scientific Trials and StudiesAurora currently has 40 clinical trials and case studies completed or in progress, 7 pre-clinical trials in progress, and 6 new partnerships with leading academic institutions in development.
Acquisitions
The Company has continued to execute on its aggressive growth strategy to build a strong business that incorporates all aspects of the cannabis value chain from greenhouse construction and cultivation to plant science research and consumer branding. During Q2 2019, Aurora has strengthened the scale of its operations, expanded its market reach, and diversified its expertise across the value chain through a number of highly strategic acquisitions and investments.
- Acquisition of ICC Labs (“ICC”)On November 22, 2018, Aurora acquired ICC Labs Inc., a leading producer and distributor of CBD and cannabinoid products in South America, a continent with over 420 million people. With over 70% market share in Uruguay and licenses to produce medical cannabis in Colombia, the acquisition of ICC Labs has established Aurora as the industry leader in South America.
- Acquisition of Whistler Medical Marijuana Corporation (“Whistler”)On January 31, 2019, the Company entered into a definitive agreement to acquire Whistler. Based in Whistler, British Columbia, Whistler is one of Canada’s most iconic cannabis brands, providing Aurora with a premium-priced and differentiated organic certified product suite to expand its medical and consumer offerings in Canada and internationally.
- Acquisition of Farmacias Magistrales S.A. (“Farmacias)On December 10th, 2018, the Company entered into a Letter of Intent to acquire Farmacias, Mexico’s first and only federally licensed importer of raw materials containing THC, with all necessary licenses, facilities, and permissions to import raw THC material, and manufacture, store, and distribute medical cannabis products containing over 1% THC. The acquisition firmly establishes Aurora’s first-mover advantage in Mexico, where more than 130 million people will have legal access to a range of Aurora’s THC containing medical cannabis products.
Investments in Canadian Consumer Retailers
- During Q2 2019, the Company increased its investment in Choom Holdings, a consumer cannabis company that has secured one of the largest retail networks in Canada, and invested in High Tide Inc, an Alberta-based, retail-focused cannabis and lifestyle accessories company.
- Through these strategic investments, Aurora further diversified its retail “pull” strategy, with additional retail opportunities across Canada.
Financing and Capital Market Activities
- New York Stock Exchange (“NYSE”) ListingOn October 23, 2018, the Company commenced trading on the NYSE under the ticker symbol ACB, exposing the company to a broader institutional investor base and larger liquidity pool.
- Australis Capital Inc. (“Australis”) Distribution On November 28, 2018, Aurora, through a selling broker, completed the sale of Australis Capital Inc. units on behalf of non-resident shareholders as a part of a spinoff of capital for Aurora Shareholders. In total, 11,222,349 units of Australis were sold in the public markets by an independent custodian, and the funds were paid to non-resident shareholders net of applicable taxes and fees.
- Private Offering of Convertible Senior NotesOn January 24, 2019, the Company issued US$345 million in aggregate principal amount of convertible senior notes. The net proceeds will be used to support Canadian and international expansion initiatives. The notes bear cash interest semi-annually at a fixed rate of 5.5% per annum, and will be convertible by holders into Aurora common shares at a conversion price of US$7.23. Upon conversion, the notes will be settled in cash, Aurora common shares or a combination of both, at Aurora’s election. The notes are unsecured and will mature by February 28, 2024.
Financial Supplement Q2 2019
Conference Call
Aurora will host a conference call today, February 11, 2019, to discuss these results. Terry Booth, Chief Executive Officer, Glen Ibbott, Chief Financial Officer, Cam Battley, Chief Corporate Officer, and Michael Singer, Chairman of the Board, will host the call starting at 6:00 p.m. Eastern time. A question and answer session will follow management’s presentation.
|
Date: |
Monday, February 11th, 2019 |
|
Time: |
6:00 p.m. Eastern Time | 4:00 p.m. Mountain Time |
|
Webcast: |
|
|
Replay: |
(416) 849-0833 or (855) 859-2056 |
|
Available until 12:00 midnight Eastern Time Monday February 18, 2019 |
|
|
Reference Number: |
2474319 |
About Aurora
Headquartered in Edmonton, Alberta, Canada with funded capacity in excess of 500,000 kg per annum and sales and operations in 23 countries across five continents, Aurora is one of the world’s largest and leading cannabis companies. Aurora is vertically integrated and horizontally diversified across every key segment of the value chain, from facility engineering and design to cannabis breeding and genetics research, cannabis and hemp production, derivatives, high value-add product development, home cultivation, wholesale and retail distribution.
Highly differentiated from its peers, Aurora has established a uniquely advanced, consistent and efficient production strategy, based on purpose-built facilities that integrate leading-edge technologies across all processes, defined by extensive automation and customization, resulting in the massive scale production of high quality product at low cost. Intended to be replicable and scalable globally, our production facilities are designed to produce cannabis of significant scale, with high quality, industry-leading yields, and low per gram production costs. Each of Aurora’s facilities is built to meet EU GMP standards, and its first production facility, the recently acquired MedReleaf Markham facility, and its wholly owned European medical cannabis distributor Aurora Deutschland, have achieved this level of certification.
In addition to the Company’s rapid organic growth and strong execution on strategic M&A, which to date includes 15 wholly owned subsidiary companies – MedReleaf, CanvasRX, Peloton Pharmaceutical, Aurora Deutschland, H2 Biopharma, Urban Cultivator, BC Northern Lights, Larssen Greenhouses, CanniMed Therapeutics, Anandia Labs, HotHouse Consulting, MED Colombia, Agropro, Borela, and ICC Labs – Aurora is distinguished by its reputation as a partner and employer of choice in the global cannabis sector, having invested in and established strategic partnerships with a range of leading innovators, including: Radient Technologies Inc. (TSXV: RTI), Hempco Food and Fiber Inc. (TSXV: HEMP), Cann Group Ltd. (ASX: CAN), Micron Waste Technologies Inc. (CSE: MWM), Choom Holdings Inc. (CSE: CHOO), Capcium Inc. (private), Evio Beauty Group (private), Wagner Dimas (private), CTT Pharmaceuticals (OTCC: CTTH), and Alcanna Inc. (TSX: CLIQ).
Aurora’s Common Shares trade on the TSX and NYSE under the symbol “ACB”, and are a constituent of the S&P/TSX Composite Index.
For more information about Aurora, please visit our investor website, investor.auroramj.com
Terry Booth, CEO
Aurora Cannabis Inc.
Read more Aurora Cannabis news here
Read all our featured articles here
Images source: Shutterstock
The Cannabis Investor is a leading media outlet for Cannabis investment opportunities and breaking industry news.
Join our text message list:
![]() USA: Text potstocks to 313131 to join
USA: Text potstocks to 313131 to join
![]() CDN: Text potstocks to 393939 to join
CDN: Text potstocks to 393939 to join
Join our email list here: http://eepurl.com/bUSa71
Follow The Cannabis Investor on Social Media
Facebook: https://www.facebook.com/MMJInvestor
Twitter: https://twitter.com/CannaInvestor
Instagram: https://www.instagram.com/thecannabisinvestor/
LinkedIn: https://www.linkedin.com/company/the-cannabis-investor/